Diffusion Defination
Diffusion is the net movement of molecules and ions from a region of their higher concentration to a region of their lower concentration down a concentration gradient, as a result of their random movement.
What is Diffusion
The molecules of a gas such as oxygen are moving about all the time. So are the molecules of a liquid or a substance such as sugar dissolved in water. As a result of this movement, the molecules spread themselves out evenly to fill all the available space (Figure).
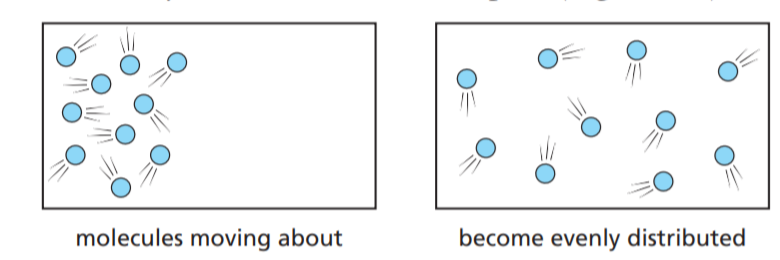
This process is called diffusion.
How are substances transported by diffusion?
One effect of diffusion is that the molecules of a gas, a liquid or a dissolved substance will move from a region where there are a lot of them (i.e. concentrated) to regions where there are few of them (i.e. less concentrated) until the concentration everywhere is the same. Figure (a) is a diagram of a cell with a high concentration of molecules (e.g. oxygen) outside and a low concentration inside.
The effect of this difference in concentration is to make the molecules diffuse into the cell until the concentration inside and outside is the same, as shown in Figure (b).
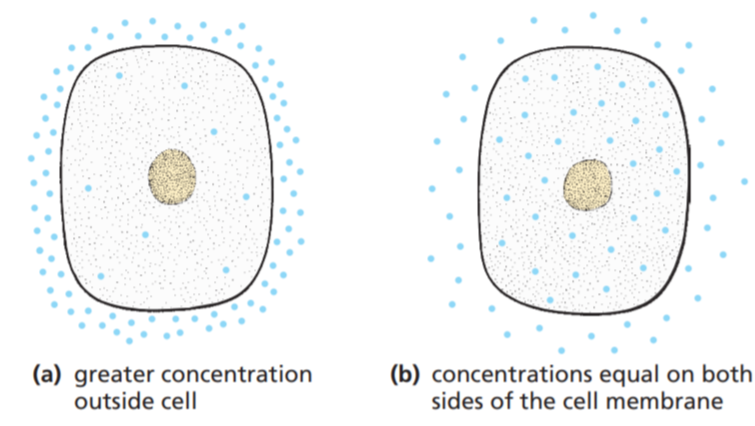
Whether this will happen or not depends on whether the cell membrane will let the molecules through. Small molecules such as water (H2O), carbon dioxide (CO2) and oxygen (O2) can pass through the cell membrane fairly easily. So diffusion tends to equalise the concentration of these molecules inside and outside the cell all the time.
When a cell uses oxygen for its aerobic respiration, the concentration of oxygen inside the cell falls and so oxygen molecules diffuse into the cell until the concentration is raised again. During tissue respiration, carbon dioxide is produced and so its concentration inside the cell increases. Once again diffusion takes place, but this time the molecules move out of the cell. In this way, diffusion can explain how a cell takes in its oxygen and gets rid of its carbon dioxide.
The importance of diffusion of gases and solutes
Gases
Most living things require a reliable source of oxygen for respiration. This moves into the organism by diffusion down a concentration gradient.
Diffusion in Animal
Small animals with a large surface area to volume ratio may obtain oxygen through their body surface. Larger animals rely on gas exchange organs such as lungs or gills, which provide a large surface area for gas exchange, and a circulatory system to transport the oxygen to all their cells.
Carbon dioxide, produced during aerobic respiration, is potentially toxic if it builds up. It is removed using the same mechanisms, again by diffusion.
Diffusion in Plant
Photosynthetic plants need carbon dioxide for making their food. This diffuses through the stomata in the leaves into the air spaces in the mesophyll, eventually reaching the palisade cells.
Oxygen, produced during photosynthesis, along with water vapour from the transpiration stream, diffuses out of the leaf through the stomata.
The rate of diffusion of water vapour depends on the temperature, humidity and wind speed. Any oxygen needed for respiration (some is generated by photosynthesis) and carbon dioxide produced (some is used up by photosynthesis) also diffuses through the stomata of the leaves.
Diffusion in Human
Nitrogen is the commonest gas in the atmosphere. (78% of the air is nitrogen.) Nitrogen gas also enters the bloodstream by diffusion, but it is not used by the body. It is an inert (unreactive) gas so, under normal circumstances, it causes no problems. However, divers are at risk.
As a diver swims deeper, the surrounding water pressure increases and this in turns raises the pressure in the diver’s air tank. An increase in nitrogen pressure in the air tank results in more nitrogen diffusing into the diver’s tissues, the amount going up the longer the diver stays at depth.
Nitrogen is not used by the body tissues, so it builds up. When the diver begins to return to the surface of the water, the pressure decreases and the nitrogen can come out of solution, forming bubbles in the blood if the diver ascends too quickly. These bubbles can block blood flow and become lodged in joints resulting in a condition called decompression sickness, or ‘the bends’.
Unless the diver rises slowly in planned stages, the effect of the nitrogen bubbles is potentially lethal and can only be overcome by rapid recompression.
Solutes
Diffusion in plant
Mineral ions in solution, such as nitrates and magnesium, are thought to diffuse across the tissues of plant roots, but most is absorbed into the roots by active transport.
Diffusion in human
In the ileum, water-soluble vitamins such as vitamin B and vitamin C are absorbed into the bloodstream by diffusion.
In the kidneys, some solutes in the renal capsule, such as urea and salts, pass back into the bloodstream by diffusion. Initially, glucose is reabsorbed by diffusion, but active transport is also involved. Dialysis machines use diffusion to remove small solutes (urea, uric acid and excess salts) from the blood.
Rates of diffusion
Molecules and ions in liquids and gases move around randomly using kinetic energy (energy from movement). The speed with which a substance diffuses through a cell wall or cell membrane will depend on temperature and many other conditions including the distance it has to diffuse, the difference between its concentration inside and outside the cell, the size of its molecules or ions and the surface area across which the diffusion is occurring.
Surface area
If 100 molecules diffuse through 1mm² of a membrane in 1 minute, it is reasonable to suppose that an area of 2mm² will allow twice as many through in the same time. Thus the rate of diffusion into a cell will depend on the cell’s surface area.
The greater the surface area, the faster is the total diffusion. Cells which are involved in rapid absorption, such as those in the kidney or the intestine, often have their ‘free’ surface membrane formed into hundreds of tiny projections called microvilli (see Figure) which increase the absorbing surface.
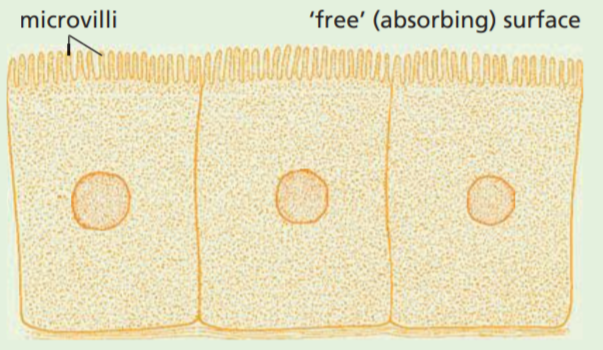
The shape of a cell will also affect the surface area. For example, the cell in Figure (a) has a greater surface area than that in Figure (b), even though they each have the same volume.

Temperature
An increase in temperature causes an increase in the kinetic energy which molecules and ions possess. This enables them to move faster, so the process of diffusion speeds up.
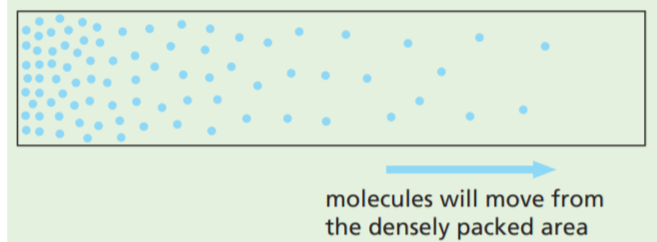
Concentration gradient
The bigger the difference in the concentration of a substance on either side of a membrane, the faster it will tend to diffuse. The difference is called a concentration gradient or diffusion gradient (Figure). If a substance on one side of a membrane is steadily removed, the diffusion gradient is maintained. When oxygen molecules enter a red blood cell they combine with a chemical (haemoglobin) which takes them out of solution. Thus the concentration of free oxygen molecules inside the cell is kept very low and the diffusion gradient for oxygen is maintained.
Distance
Cell membranes are all about the same thickness (approximately 0.007 µm) but plant cell walls vary in their thickness and permeability. Generally speaking, the thicker the wall, the slower the rate of diffusion. When oxygen diffuses from the alveoli of the lungs into red blood cells, it has to travel through the cell membranes of the alveoli, the blood capillaries and the red blood cells in addition to the cytoplasm of each cell. This increased distance slows down the diffusion rate.
Size of molecules or ions
In general, the larger the molecules or ions, the slower they diffuse. However, many ions and molecules in solution attract water molecules around them and so their effective size is greatly increased. It may not be possible to predict the rate of diffusion from the molecular size alone.
Controlled diffusion
Although for anyone substance, the rate of diffusion through a cell membrane depends partly on the concentration gradient, the rate is often faster or slower than expected. Water diffuses more slowly and amino acids diffuse more rapidly through a membrane than might be expected. In some cases, this is thought to happen because the ions or molecules can pass through the membrane only by means of special pores. These pores may be few in number or they may be open or closed in different conditions.
In other cases, the movement of a substance may be speeded up by an enzyme working in the cell membrane. So it seems that ‘simple passive’ diffusion, even of water molecules, may not be so simple or so passive after all where cell membranes are concerned.
When a molecule gets inside a cell there are a great many structures and processes which may move it from where it enters to where it is needed. Simple diffusion is unlikely to play a very signifi cant part in this movement.



